Back: Go to Home Page or Go to Chemistry MCQS
– UV – Visible Organic Chemistry Questions and Answers
Q1. This set of Organic Chemistry Multiple Choice Questions & Answers (MCQs) focuses on “UV – Visible Spectroscopy”.
1. What is the wavelength range for UV spectrum of light?
a) 400 nm – 700 nm
b) 700 nm to 1 mm
c) 0.01 nm to 10 nm
d) 10 nm to 400 nm
Answer: d
Explanation: Ultraviolet (UV) is an electromagnetic radiation with a wavelength from 10 nm to 400 nm, shorter than that of visible light but longer than X-rays (the visible region fall between 380-750 nm and X- rays region fall between 0.01 to 10nm).
_____________________________________________________________________________
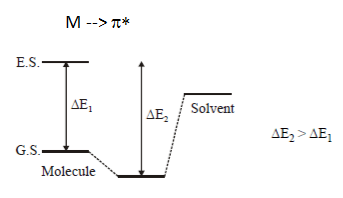
Q2.Which of the following comparison is correct for solvent shift on the n –>π* transition of acetone?
a) H20 = CH30H = C2H50H = CHC13 = C6H14
b) H20 > CH30H > C2H50H > CHC13 > C6H14
c) H20 < CH30H < C2H50H < CHC13 < C6H14
d) H20 > CH30H < C2H50H < CHC13 < C6H14
Answer: c
Explanation: H-bonding with ground state in n–> π* results in increase in energy gap & decrease in wavelength.
And as polar solvents show strong H-bonding. So, the correct option is H20 < CH30H < C2H50H < CHC13 < C6H14.
organic-chemistry-questions-answers-uv-visible-spectroscopy-q2
_____________________________________________________________________________
_____________________________________________________________________________
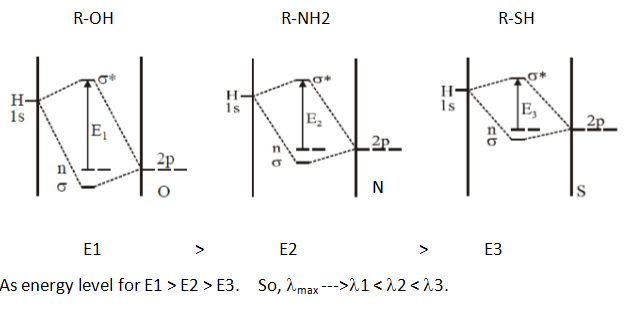
Q3. What is the correct order of λmax for n –> σ* transition?
a) R-OH > R-NH2 > R-SH
b) R-OH < R-NH2 < R-SH
c) R-OH > R-SH > R-NH2
d) R-OH < R-SH < R-NH2
Answer: b
Explanation: According to molecular orbital energy diagram for R-OH, R-NH2, R-SH (shown below), energy level will decrease respectively so λmax will increase.
_____________________________________________________________________________
Q4. What is the correct order of λmax for n –> π* transition for the R-CN, R-NO2, and R-N=N-R?
a) R-CN < R-NO2 < R-N=N-R
b) R-CN = R-NO2 = R-N=N-R
c) R-CN > R-NO2 > R-N=N-R
d) R-CN > R-NO2 < R-N=N-R
a) R-CN < R-NO2 < R-N=N-R
b) R-CN = R-NO2 = R-N=N-R
c) R-CN > R-NO2 > R-N=N-R
d) R-CN > R-NO2 < R-N=N-R
Answer: a
Electronegative difference order: 3 < 2 < 1
If different is less than energy gap between n –> π* is also less hence more wavelength.
So, energy gap between n –> π* 3 < 2 < 1
λmax–> 3 > 2 > 1
If different is less than energy gap between n –> π* is also less hence more wavelength.
So, energy gap between n –> π* 3 < 2 < 1
λmax–> 3 > 2 > 1
_____________________________________________________________________________
Q5. What is the correct order of λmax for π –> π* transition for the following three compounds?
a) R-C=C-R > R2C = CR2 > R-CHO
b) R-C=C-R < R2C = CR2 < R-CHO
c) R-C=C-R = R2C = CR2 = R-CHO
d) R-C=C-R < R2C = CR2 >R-CHO
a) R-C=C-R > R2C = CR2 > R-CHO
b) R-C=C-R < R2C = CR2 < R-CHO
c) R-C=C-R = R2C = CR2 = R-CHO
d) R-C=C-R < R2C = CR2 >R-CHO
Answer: b
Explanation: MO diagram (C = C), (C = C) & (C = O) and shows the energy gap between π – π*, increases among these three molecules. So λmax will give the reverse trend.
R-C=C-R < R2C = CR2 < R-CHO
_____________________________________________________________________________
Q6. What is the correct order of λmax for n–> π* transition for the following three compounds?
a) RCOOH > RCOOR’ > RCONH2
b) RCOOH = RCOOR’ = RCONH2
c) RCOOH = RCOOR’ < RCONH2
d) RCOOH = RCOOR’ > RCONH2
a) RCOOH > RCOOR’ > RCONH2
b) RCOOH = RCOOR’ = RCONH2
c) RCOOH = RCOOR’ < RCONH2
d) RCOOH = RCOOR’ > RCONH2
Answer: b
Explanation:
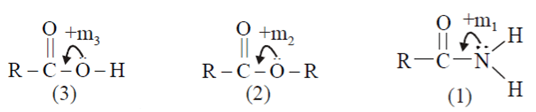
As we can see from above diagram: +m1 > +m2; +m3
Here since mesomeric effect shows Bathochromic shift: i.e. a change of spectral band position in the absorption, reflectance, transmittance, or emission spectrum of a molecule to a longer wavelength (lower frequency)
Here, λmax –> 3 ≈ 2 < 1.
_____________________________________________________________________________
Q7. Which of the following is the correct order of λmax for n–> π* transition for the given compounds?
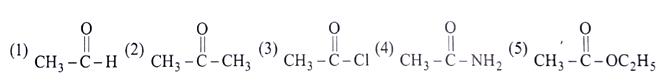
a) 1>2>3>4>5
b) 1<2<3<4<5
c) 1>2<3>4<5
d) 1>2>3<4>5
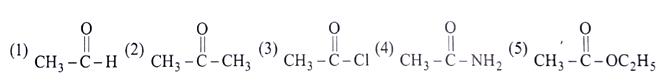
a) 1>2>3>4>5
b) 1<2<3<4<5
c) 1>2<3>4<5
d) 1>2>3<4>5
Answer: a
Explanation: λmax depends upon +m & I effect, which will lead to a shift to longer wavelength accompanied by increased intensity of absorption represents an electronic level (considering inductive and mesomeric effect) of the ground and excited states and thus a decrease in transition energy.
The correct order of λmax is 1 > 2 > 3 > 4 > 5.
_____________________________________________________________________________
Q8. The ultraviolet spectrum of benzonitrile shows a primary absorption band at 224 nm. If a solution of benzonitrile in water, with a concentration of 1x 10-4 molar, is examined at a wavelength of 224 nm, the absorbance is determined to be 1.30. The cell length is 1 cm.
What is the molar absorptivity of this absorption band?
a) 2.3 x 104
b) 3.3 x 104
c) 1.3 x 104
d) 4.3 x 104
What is the molar absorptivity of this absorption band?
a) 2.3 x 104
b) 3.3 x 104
c) 1.3 x 104
d) 4.3 x 104
Answer: c
.Explanation:
Benzonitrile in water with:C = 1 x 10-4 M
A = 1.30
l = 1 cm
We know, A = ℇCl
ℇ = A/C I = 1.30/ 1X 10-4 X 1 = 1.34 X 104.
_____________________________________________________________________________
Q9. The ultraviolet spectrum of benzonitrile shows a secondary absorption band at 271 nm. If a solution of benzonitrile in water, with a concentration of 1×10-4 molar solution is examined at 271 nm, what will be the absorbance reading (ℇ = 1000) and what will be the intensity ratio, IO/I, respectively?
a) 0.1, 1.26
b) 0.2, 2.26
c) 0.3, 3.26
d) 0.4, 4.26
a) 0.1, 1.26
b) 0.2, 2.26
c) 0.3, 3.26
d) 0.4, 4.26
Answer: a
Explanation: A = ℇCl and ℇ = 1000 (Given)
A = 1000 x 1 x10-4 x 1
A = 0.1
Also, A = log (IO/l)
0.1 = log (IO/I)
=> IO/I = 1.26.
_____________________________________________________________________________
Q10. Which of the following structural formula that is consistent with the following observations: An acid, C7H4O2Cl2 shows a UV maximum of 242 nm?
a)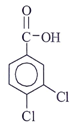
b)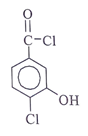
c)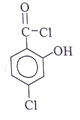
d)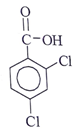
a)

b)

c)

d)

Answer: c
Explanation: λx at 242 nm represents it is acid chloride. -OH at ortho position show +m effect & hence λmax increases to 242 nm.




0 comments:
Post a Comment